
 The disintegration tester called DISI is the universal, all-knowing instrument for those who carry out disintegration tests in the pharmaceutical industry. This instrument is capable of recording the complete disintegration profile of the products to be tested. The DISI instrument, with its magnetic basket suspensions and contact-free signal transfer, provides the simplicity of the tests and the speed of the basket changeover during the subsequent cleaning processes. The DISI instruments are designed with 1, 2, 3 or 4 baskets. The baskets are of magnetic suspension, in this way they provide automatic centering and a very easy changeover. The detection of the changes in the process of disintegration is done with the help of the wire rings built in the disks that are placed above the tablets and based on the magnetic induction principle. The signal transfer is also based on magnetic principle without electrical contacts and the potentially accompanying corrosion. The temperature sensor placed in the basket holder, the temperature of the medium used for the disintegration testing can be constantly followed and documented.
The disintegration tester called DISI is the universal, all-knowing instrument for those who carry out disintegration tests in the pharmaceutical industry. This instrument is capable of recording the complete disintegration profile of the products to be tested. The DISI instrument, with its magnetic basket suspensions and contact-free signal transfer, provides the simplicity of the tests and the speed of the basket changeover during the subsequent cleaning processes. The DISI instruments are designed with 1, 2, 3 or 4 baskets. The baskets are of magnetic suspension, in this way they provide automatic centering and a very easy changeover. The detection of the changes in the process of disintegration is done with the help of the wire rings built in the disks that are placed above the tablets and based on the magnetic induction principle. The signal transfer is also based on magnetic principle without electrical contacts and the potentially accompanying corrosion. The temperature sensor placed in the basket holder, the temperature of the medium used for the disintegration testing can be constantly followed and documented.
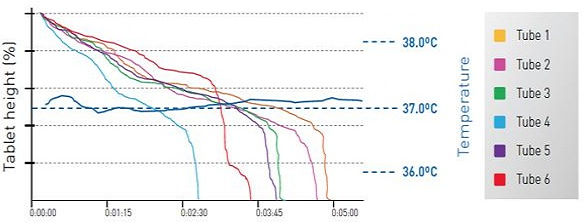
Tablets, capsules, tablets with multistage disintegration can be tested and also the disintegration of products about which the electrical endpoint indicators provide false results due to the non-disintegrated scrap. The control of the DISI is carried out by the PH21 software, which corresponds to the requirements of the 21 CFR Part 11 and a wide range of password protection, statistics calculation and disintegration curves belong to it.
Specifications:
- conformity: USP and EP
- stroke length: 55 mm
- frequency: 30 ± 0.1 stroke/min
- temperature: from room temperature to 40 oC
- detection: magnetic induction
- signal coupling: electromagnetic, without electric cables and contacts
- configuration: 1, 2, 3 or 4 basket
- baskets: 6 position or position (bolus)
- basket movement: each basket independent
- basket positioning: magnetic, automatic
- control: PH21 Windows software

